FDA Application was Filed
Biop filled the 510k application for the colposcopic device. This first application submitted by Biop is comprised of approx. 800...


IBM shares news about Biop on company website
/ Yoast SEO plugin. WordPress Popular Posts v3.3.4 End WordPress Popular Posts v3.3.4 ================================================...


BIOP MEDICAL @ MEDICA TRADEFAIR
Nov. 13-16 in Dusseldorf, Germany Biop Medical launched the technology and device for early screening of cervical cancer at the...


Biop Medical at Healthcare Business News
https://www.dotmed.com/news/story/4016 6


Our New Film
To understand better what we do. Watch our new company film
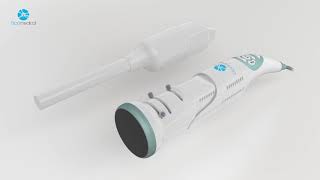

News Realese from Biop Medical Nov 2017
BIOP MEDICAL LAUNCHES CERVICAL CANCER DIAGNOSIS TECH AT MEDICA 2017 RAMAT GAN, Israel, November 7 – Biop Medical announced it will...


PRODUCT LAUNCH AT THE MEDICA TRADE FAIR
Next month, 13-16 November,we will be presenting the Alpha version of our product for the first time at the prestigious Medica tradefair...






























